Research Goals
Our goal is to understand the molecular mechanisms of neuronal development and regeneration, and to utilize gained knowledge in developing translational approaches for repairing injured central nervous system (CNS) circuits.
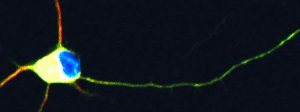
An image of immature retinal ganglion cell neuron from Dr. Trakhtenberg's research, adapted for a cover page of the International Review of Neurobiology volume on Axon Growth and Regeneration (Goldberg & Trakhtenberg, Eds, 2012, Vol 106: Academic Press).
NEWS
May, 2026
A research article from our lab was published in Experimental Neurology: Experimental upregulation of Lancl1 promotes axon regeneration after optic nerve injury in vivo. It was selected for a cover page! This study was funded by the National Institutes of Health's Eye Institute (NEI) R01 grant.
April, 2026
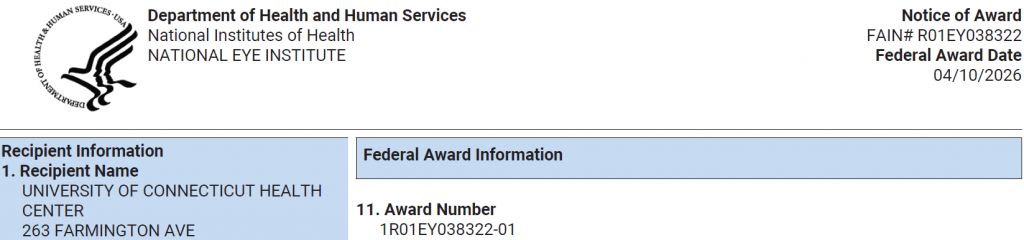
The National Institutes of Health's Eye Institute (NEI) awarded Dr. Trakhtenberg an R01 grant to support our lab’s neurotherapeutic R&D science for optic neuropathies and other CNS diseases.
March, 2026
A research article from our lab was published in Molecular Therapy Nucleic Acids: Vacuolar ATPase subunit Atp6v0c transgene promotes neuroprotection and long-distance axon regeneration in injured retinal ganglion neurons. This study was funded by the National Institutes of Health's Eye Institute (NEI) R01 grant.
January, 2025
A Chapter, Single-cell transcriptomics-enabled advances in experimental optic nerve axon regeneration research, by Dr. Trakhtenberg, was published in Bhattacharya. S.K. (Ed.): Proteomics, Multi-Omics and Systems Biology in Optic Nerve Regeneration, 2025; Elsevier.
December, 2024
A recent discovery from our lab was covered in several news outlets: EyeWire, UConnToday, OphthalmologyBreakingNews, PatientWorthy, NIH!
 Left-to-right: Dr. Bruce Rheaume (now a physician-scientist at Dartmouth Medical Center), Dr. Agnieszka Lukomska (now a professor at the University of Warsaw), Dr. Feliks (Ephraim) Trakhtenberg (Principal Investigator), Anja Kearney (undergraduate honors student), Mahit Gupta (undergraduate honors student), and Matthew Frost (PhD candidate).
Left-to-right: Dr. Bruce Rheaume (now a physician-scientist at Dartmouth Medical Center), Dr. Agnieszka Lukomska (now a professor at the University of Warsaw), Dr. Feliks (Ephraim) Trakhtenberg (Principal Investigator), Anja Kearney (undergraduate honors student), Mahit Gupta (undergraduate honors student), and Matthew Frost (PhD candidate).
September, 2024
A research article from our lab was published in Experimental Neurology: Augmenting fibronectin levels in injured adult CNS promotes axon regeneration in vivo. It was selected for a cover page! This study was funded by the National Institutes of Health's Eye Institute (NEI) R01 grant. Was covered in the news Nerve Regrowth in Sight (by Kim Krieger,published in UConn Today), including by the NIH National Eye Institute, which funded these studies, as well as by other research news outlets.
February, 2024
A research article from our lab was published in Neuroscience Letters: Upregulation of developmentally-downregulated miR-1247-5p promotes neuroprotection and axon regeneration in vivo. This study was funded by the National Institutes of Health's Eye Institute (NEI) R01 grant.
January, 2024
A research article from our lab was published in Experimental Neurology: Nfe2l3 promotes neuroprotection and long-distance axon regeneration after injury in vivo. It was selected for a cover page! This study was funded by the National Institutes of Health's Eye Institute (NEI) R01 grant. Was covered in the news Seeing a path to nerve regeneration (by Kim Krieger, published in UConn Today), including by the NIH National Eye Institute, which funded these studies, as well as by other research news outlets.
October, 2023
A research article from our lab was published in Experimental Neurology, Experimental upregulation of developmentally downregulated ribosomal protein large subunits 7 and 7A promotes axon regeneration after injury in vivo, and was covered in the news UConn Health Researchers Find that Youthful Proteins Help Nerves Regrow (by Kim Krieger, published in UConn Today), including by the NIH National Eye Institute, which funded these studies, as well as by other research news outlets.
September, 2023
Dr. Trakhtenberg contributed to the editing of a review article, recently published in Molecular Neurodegeneration, as a member of the RReSTORe Consortium, which coauthored the article: Retinal ganglion cell repopulation for vision restoration in optic neuropathy: a roadmap from the RReSTORe Consortium.
July, 2023
A perspective/review article by Dr. Trakhtenberg was published in Neural Regeneration Research: Premature axon-oligodendrocyte interaction contributes to stalling of experimental axon regeneration after injury to the white matter.
June, 2023
UConn Health press release on recent discoveries from our lab, New insights into how axons grow could lead to effective therapies for blindness, paralysis (by Kim Krieger, published in UConn Today), was covered in research news of the NIH National Eye Institute, which funded these studies, and in multiple news outlets:
May, 2023
UConn undergraduate student, Ashiti Damania, who did her honors thesis and was a research assistant in our lab over the past 3 years through graduation this Spring, won a prestigious University Scholar Award and co-authored 4 published research articles.
May, 2023
Graduate students from our lab presented three posters on their latest discoveries at the Department of Neuroscience Annual Retreat, which featured keynote speaker Dr. Marina Picciotto from Yale University.

May, 2023
Two research articles from our lab were published in Brain Research: Transcriptomic profiling of retinal cells reveals a subpopulation of microglia/macrophages expressing Rbpms marker of retinal ganglion cells (RGCs) that confound identification of RGCs and Experimental gene expression of developmentally downregulated Crmp1, Crmp4, and Crmp5 promotes axon regeneration and retinal ganglion cell survival after optic nerve injury.
April, 2023
Two research articles from our lab were published in Development: Post-injury born oligodendrocytes incorporate into the glial scar and contribute to the inhibition of axon regeneration and Pten inhibition dedifferentiates long-distance axon-regenerating intrinsically photosensitive retinal ganglion cells and upregulates mitochondria-associated Dynlt1a and Lars2. These discoveries were highlighted and selected for a published interview, "The people behind the papers".
April, 2023
Graduate student, Matthew Frost, presented a poster on our lab's latest discoveries at the Vision Injury Research Forum (VIRF), co-hosted by The Department of Defense (DOD), Vision Research Program (VRP), and Vision Center of Excellence (VCE).
March, 2023
Graduate student, Matthew Frost, won Lightning Talk 2nd place award on presentation of our lab's latest discoveries at the 8th Military Vision Symposium (MVS): Innovation and Unmet Needs for Ocular Injury, Boston, MA.
November, 2022
Postdoctoral fellow, Agnieszka Lukomska, and graduate student, Jian Xing, won Travel Awards and presented two posters on our lab's latest discoveries at the Society for Neuroscience conference, San Diego.
April, 2022
Graduate student, Bruce Rheaume, defended his Ph.D. thesis, and returned to the medical school to complete M.D./Ph.D. training.
July, 2022
Dr. Trakhtenberg's research recognized in the National Glaucoma Research Report by the BrightFocus Foundation, which funds his research on investigating novel gene therapy approach towards developing neuroregenerative treatments for restoring vision after certain types of glaucoma and other types of optic neuropathies.
October, 2021
Preprint of a research article from our lab is now available at bioRxiv. The article includes a link to the scRNAseq-based Subtypes Gene Browser for adult uninjured and injured RGCs (the latter re-analyzed using next generation algorithms from raw data by Tran et al. 2019): Retroactive analysis of single cell transcriptome profiles using next-generation algorithms revised the identification of several resilient retinal ganglion cell types
September, 2021
A research article from our lab was published in Neuroscience Letters: Developmentally upregulated Transcriptional Elongation Factor A like 3 suppresses axon regeneration after optic nerve injury. Covered in the news by the BrightFocus Foundation.
November, 2020
A research article in collaboration with Royce Mohan’s lab was published in the Journal of Neuroscience Research Corneal nonmyelinating Schwann cells illuminated by single-cell transcriptomics and visualized by protein biomarkers
July, 2019
Dr. Trakhtenberg received UConn Health's Faculty Spotlight recognition for “Research Excellence”, which includes being awarded an NIH grant for his novel research approach to restoring vision loss.
June, 2019
Bruce Rheaume, MD/PhD Candidate, won Biomedical Science Program Mentorship Award for outstanding mentorship by a student in the biomedical science program.
May, 2019
Dr. Trakhtenberg was awarded a 2 million 5-year R01 grant by the National Institutes of Health's Eye Institute (NEI) to investigate how small non-coding RNAs regulate retinal ganglion cell maturation and to utilize their potential for regenerating the optic nerve axons damaged in optic neuropathies.
August, 2018
Our lab in collaboration with JAX published in Nature Communications first molecular classification of retinal ganglion cells into subtypes, covered in the news.
June, 2018
Dr. Trakhtenberg and Dr. Wu were awarded a Research Excellence Program Grant by The Office of the Vice President for Research, UConn School of Medicine and School of Dental Medicine.
June, 2018
Our lab published in Scientific Reports that the extent of non-atrophic extra-axonal tissue damage determines the success of experimental axon regeneration targeting neuronal intrinsic mechanisms.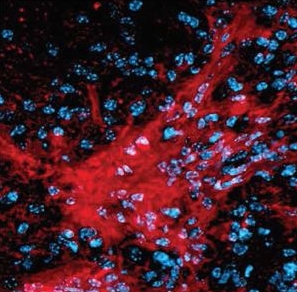
May, 2018
Dr. Trakhtenberg and Dr. Rouge were awarded a PITCH Seed Award Grant by the PITCH, featured in UConnToday news.
March, 2018
Dr. Trakhtenberg has received Artificial Intelligence Molecular Screen (AIMS) Award, Atomwise Inc (San Francisco, CA) .
November, 2017
An MD-PhD student, Bruce Rheaume, presented a poster from the lab, "Axotomized adult retinal ganglion cells stimulated by extrinsic cues in a permissive environment survive and regenerate axons", at the international annual meeting of the Society for Neuroscience (SFN) in Washington, DC.
September, 2017
Dr. Trakhtenberg’s awards and his lab were featured in UConnToday news.

June, 2017
Dr. Trakhtenberg (lead PI) and Dr. Crocker (co-PI) were awarded a seed grant by the Connecticut Institute for the Brain and Cognitive Sciences (IBaCS). The grant will fund a research project to test a novel hypothesis regarding why axonal connections, through which neurons in the brain communicate with each other over long distances, do not regenerate after traumatic or stroke injury.
April, 2017
Dr. Trakhtenberg was awarded a research grant by the BrightFocus Foundation under the National Glaucoma Research Program. The grant will fund a research project aimed at investigating novel gene therapy approach towards developing neuroregenerative treatments for restoring vision after angle-closure glaucoma and other types of optic neuropathies, which lead to complete or partial blindness.
March, 2017
Dr. Trakhtenberg was selected by the New York Academy of Sciences and the Japan Agency for Medical Research and Development to participate at the Interstellar Initiative for “the world's most promising Early Career Investigators”, where along with a collaborator, Dr. Kumiko Hayashi, they won First Place Award for a research solution proposal in the field of neuroscience.

CURRENT OPENINGS
Postdoctoral Fellow
Graduate Rotation Student
Projects in the lab revolve around fundamental questions in neuronal development and regeneration in the CNS. We integrate cutting edge molecular, biochemical, genetic, bioinformatics, and translational approaches, which involve:
- Histology and neuroanatomical analysis using confocal microscopy
- Rodent CNS in vivo injury models and gene therapy
- Neuronal cell culture and transfection
- Next-generation sequencing and neuro-bioinformatics