Research
The primary focus of my laboratory is studies of protein structure, dynamics and interactions using structural biology methods, including nuclear magnetic resonance (NMR) spectroscopy and X-ray crystallography. We make use of cutting-edge TROSY-NMR techniques that allow to access structural dynamics and interactions of protein assemblies with molecular weights of up to 1 MDa, opening an avenue for deciphering molecular mechanisms of their action.
DNA damage tolerance and cancer:
The underlying cause of cancer is spontaneous mutations introduced to genomic DNA. Reactive products of cellular metabolism and external genotoxic agents cause persistent DNA damage, which is constantly removed through various DNA repair mechanisms. It is unavoidable, however, that some DNA modifications (lesions) persist into S-phase, creating blocks for progression of the DNA replication machinery. To circumvent this problem organisms in all kingdoms of life have evolved DNA damage tolerance pathways, employing specialized enzymes that bypass DNA lesions while temporarily leaving DNA damage unrepaired. The vast majority of mutations are introduced in the genome by enzymes of error-prone branch of DNA damage tolerance - translesion DNA synthesis (TLS). Genetic changes that ensue as a result of TLS are at the root of the onset of cancer and the development of various resistance mechanisms displayed by relapsed tumors, which represents a major problem for treatment of some types of cancer, including ovarian and lung. Our research is aimed at obtaining a detailed atomic-resolution picture of structure, dynamics and interactions of proteins and protein assemblies involved in DNA damage tolerance pathways that will aid the development of new strategies for cancer therapy.
Protein folding and dynamics:
Intermediate and transition states of biomolecular processes represent a paradigm of functionally important structure in biology. For example, protein self-assembly involves the formation of partially folded and misfolded protein states prone to aggregation implicated in a number of human disorders, including type-II diabetes, Alzheimer's and Parkinson's diseases. Although the characterization of such species can provide vital clues about the mechanisms of the underlying processes, it is extremely challenging to examine such states because they are populated at low levels and are not readily isolated. One of the research directions in my laboratory is studies of intermediate and transition states of protein folding and binding using novel NMR relaxation dispersion methodology.
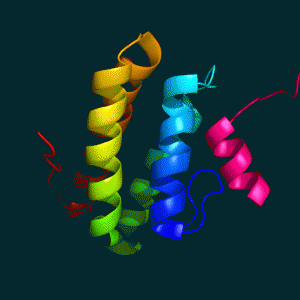 REV1 C-terminal Domain
REV1 C-terminal Domain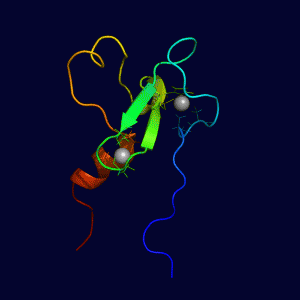 SHPRH PHD Domain
SHPRH PHD Domain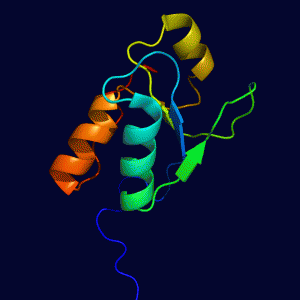 REV1 BRCT Domain
REV1 BRCT Domain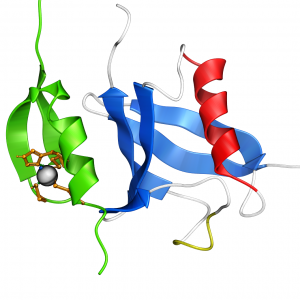 Rad18 UBZ Domain in Complex with Ubiquitin
Rad18 UBZ Domain in Complex with Ubiquitin
Publications
2026
USP1 inhibition promotes RAD18-dependent PCNA degradation and BRCA1 synthetic lethality
Ashton NW, Ravindranathan R, Korchak EJ, Somuncu OS, Zambrano GA, Mukkavalli SV, Asada S, Korzhnev DM, Bezsonova I, D’Andrea AD.
bioRxiv. 2026 May 10:2026.02.25.708068.
doi: 10.64898/2026.02.25.708068
PMID: 42146504 (preprint)
2025
Long-range destabilizing effects of mutations at the Escherichia coli β clamp dimer interface
Liriano ML, Berger MB, Nguyen K, Korzhnev DM, Cisneros GA, Beuning PJ.
Biochemistry. 2025 Jul 15;64(14):3126–3136.
doi: 10.1021/acs.biochem.5c00135
PMID: 40574562
Functional asymmetry in processivity clamp proteins
Mahdi S, Beuning PJ, Korzhnev DM.
Biophys J. 2025 May 20;124(10):1549–1561.
doi: 10.1016/j.bpj.2025.04.014
PMID: 40247618
Activation dynamics of ubiquitin-specific protease 7
Valles GJ, Korchak EJ, Geddes-Buehre DH, Jaiswal N, Korzhnev DM, Bezsonova I.
PNAS. 2025 May 27;122(21):e2426632122.
doi: 10.1073/pnas.2426632122
PMID: 40397674
Divide-and-conquer strategy for NMR studies of the E. coli γ-clamp loader complex
Mahdi S, Semenova IV, Bezsonova I, Beuning PJ, Korzhnev DM.
J Biomol NMR. 2025;79(4):243–257.
doi: 10.1007/s10858-025-00471-0
PMID: 40694188
Differences in structure, dynamics, and zinc coordination between isoforms of human ubiquitin ligase UBE3A
Bregnard TA, Fairchild D, Chen X, Erlandsen H, Tarasov SG, Walters KJ, Korzhnev DM, Bezsonova I.
J Biol Chem. 2025 Feb;301(2):108149.
doi: 10.1016/j.jbc.2024.108149
PMID: 39742997
REV1 inhibition enhances trinucleotide repeat mutagenesis
Siegel A, Almstead D, Kothandaraman N, Reich J, Lamkin E, Victor JA, Grover A, Ikeh K, Koval H, Crompton A, Jang H, Lee H, Del Rio Guerra R, Korzhnev DM, Hadden MK, Hong J, Zhou P, Chatterjee N.
bioRxiv. 2025 Sep 16:2025.09.11.675234.
doi: 10.1101/2025.09.11.675234
PMID: 41000971 (preprint)
REV1 inhibition enhances trinucleotide repeat mutagenesis
Siegel A, Almstead D, Kothandaraman N, Reich J, Lamkin E, Victor J, Grover A, Ikeh K, Koval H, Crompton A, Jang H, Lee H, Del Rio-Guerra R, Korzhnev D, Hadden MK, Hong J, Zhou P, Chatterjee N.
Open Biol. 2025 Dec 1;15(12):250234.
doi: 10.1098/rsob.250234
PMID: 41537824
2024
Probing hot spots of protein-protein interactions mediated by the safety-belt region of REV7
Dash RC, Arianna GA, Patel SM, Rizzo AA, Harrahill NJ, Korzhnev DM, Hadden MK.
Structure. 2024 Nov 7;32(11):2134–2146.e3.
doi: 10.1016/j.str.2024.09.007
PMID: 39366370
Backbone NMR resonance assignments of stabilized E. coli β clamp
Mahdi S, Lim S, Bezsonova I, Beuning PJ, Korzhnev DM.
Biomol NMR Assign. 2024;18(2):293–297.
doi: 10.1007/s12104-024-10202-5
PMID: 39269602
Protein assemblies in translesion synthesis
Arianna GA, Korzhnev DM.
Genes (Basel). 2024;15(7):832.
doi: 10.3390/genes15070832
PMID: 39062611
Lead compound profiling for small molecule inhibitors of REV1-CT/RIR interaction
Zaino AM, Dash RC, James SJ, MacGilvary N, Crompton A, McPherson KS, Stanzione M, Korzhnev DM, Dyson NJ, Chatterjee N, Cantor SB, Hadden MK.
Bioorg Med Chem. 2024 May 15;106:117755.
doi: 10.1016/j.bmc.2024.117755
PMID: 38749343
Effects of xylanase A double mutation on substrate specificity and structural dynamics
MacDonald ME, Wells NGM, Hassan BA, Dudley JA, Walters KJ, Korzhnev DM, Aramini JM, Smith CA.
J Struct Biol. 2024 Jun;216(2):108082.
doi: 10.1016/j.jsb.2024.108082
PMID: 38438058
2023
Conformational exchange at a C2H2 zinc-binding site facilitates redox sensing by PML protein
Bregnard TA, Fairchild D, Erlandsen H, Semenova IV, Szczepaniak R, Ahmed A, Weller SK, Korzhnev DM, Bezsonova I.
Structure. 2023 Sep 7;31(9):1086–1099.e6.
doi: 10.1016/j.str.2023.06.014
PMID: 37473756
Backbone and ILV methyl NMR assignments of Rev7/Rev3 complexes
Arianna GA, Geddes-Buehre DH, Korzhnev DM.
Biomol NMR Assign. 2023 Jun;17(1):107–114.
doi: 10.1007/s12104-023-10128-4
PMID: 37129702
Activation Dynamics of Ubiquitin Specific Protease 7
Valles GJ, Jaiswal N, Korzhnev DM, Bezsonova I.
bioRxiv. 2023 Jan 12:2023.01.11.523550.
doi: 10.1101/2023.01.11.523550
PMID: 36711877 (preprint)
Evolution of Rev7 interactions in eukaryotic TLS DNA polymerase Polζ
McPherson KS, Rizzo AA, Erlandsen H, Chatterjee N, Walker GC, Korzhnev DM.
J Biol Chem. 2023;299(2):102859.
doi: 10.1016/j.jbc.2022.102859
PMID: 36592930
2022
DNA sequence specificity reveals HLTF HIRAN role in trinucleotide repeat recognition
Dusek CO, Dash RC, McPherson KS, Calhoun JT, Bezsonova I, Korzhnev DM, Hadden MK.
Biochemistry. 2022.
doi: 10.1021/acs.biochem.2c00027
PMID: 35608245
NMR resonance assignments of USP7 catalytic domain
Valles G, Pozhidaeva A, Korzhnev DM, Bezsonova I.
Biomol NMR Assign. 2022;16(2):197–203.
doi: 10.1007/s12104-022-10079-2
PMID: 35536398
ILV methyl NMR assignments of E. coli β clamp
Lim S, Mahdi S, Beuning PJ, Korzhnev DM.
Biomol NMR Assign. 2022;16(2):317–323.
doi: 10.1007/s12104-022-10097-0
PMID: 35687262
Architecture of two metal-binding sites in prolactin
Vang J, Pustovalova Y, Korzhnev DM, Gorbatyuk O, Keeler C, Hodsdon ME, Hoch JC.
Biophys J. 2022;121(7):1312–1321.
doi: 10.1016/j.bpj.2022.02.024
PMID: 35192840
2021
REV1 inhibition enhances radioresistance and autophagy
Ikeh KE, Lamkin EN, Crompton A, Deutsch J, Fisher KJ, Gray M, Argyle DJ, Lim WY, Korzhnev DM, Hadden MK, Hong J, Zhou P, Chatterjee N.
Cancers (Basel). 2021;13(21):5290.
doi: 10.3390/cancers13215290
PMID: 34771454
Targeting protein-protein interactions in DNA damage response
McPherson KS, Korzhnev DM.
RSC Chem Biol. 2021;2(4):1167–1195.
doi: 10.1039/d1cb00101a
PMID: 34458830
2019
Rev1-Polζ mutasome structure and inhibition
Rizzo AA, Korzhnev DM.
Enzymes. 2019;45:139–181.
doi: 10.1016/bs.enz.2019.07.001
PMID: 31627876
Virtual pharmacophore screening for Rev1 inhibitors
Dash RC, Ozen Z, McCarthy KR, Chatterjee N, Harris CA, Rizzo AA, Walker GC, Korzhnev DM, Hadden MK.
ChemMedChem. 2019;14(17):1610–1617.
doi: 10.1002/cmdc.201900307
PMID: 31361935
Dynamics of β-clamp dimer interface
Koleva BN, Gokcan H, Rizzo AA, Lim S, Jeanne Dit Fouque K, Choy A, Liriano ML, Fernandez-Lima F, Korzhnev DM, Cisneros GA, Beuning PJ.
Biophys J. 2019;117(3):587–601.
doi: 10.1016/j.bpj.2019.06.035
PMID: 31349986
2018
Rev7 dimerization in TLS complex assembly
Rizzo AA, Vassel FM, Chatterjee N, D’Souza S, Li Y, Hao B, Hemann MT, Walker GC, Korzhnev DM.
PNAS. 2018;115:E8191–E8200.
doi: 10.1073/pnas.1801149115
PMID: 30111544
Small molecules disrupting Rev1-CT/RIR interaction
Ozen Z, Dash RC, McCarthy KR, Chow SA, Rizzo AA, Korzhnev DM, Hadden MK.
Bioorg Med Chem. 2018;26(14):4301–4309.
doi: 10.1016/j.bmc.2018.07.029
PMID: 30037752
Structural scaffold identification for Rev1 targeting
Dash RC, Ozen Z, Rizzo AA, Lim S, Korzhnev DM, Hadden MK.
J Chem Inf Model. 2018;58(11):2266–2277.
doi: 10.1021/acs.jcim.8b00535
PMID: 30289707
2017
Protein folding by NMR
Zhuravleva A, Korzhnev DM.
Prog Nucl Magn Reson Spectrosc. 2017;100:52–77.
doi: 10.1016/j.pnmrs.2016.10.002
PMID: 28552172
Small-molecule TLS inhibitors
Sail V, Rizzo AA, Chatterjee N, Dash RC, Ozen Z, Walker GC, Korzhnev DM, Hadden MK.
ACS Chem Biol. 2017;12(7):1903–1912.
doi: 10.1021/acschembio.6b01144
PMID: 28541665
Early folding nucleation events
Kukic P, Pustovalova Y, Camilloni C, Gianni S, Korzhnev DM, Vendruscolo M.
J Am Chem Soc. 2017;139:6899–6910.
doi: 10.1021/jacs.7b01540
PMID: 28401755
Clamp loader δ-subunit assignments
Alyami EM, Rizzo AA, Beuning PJ, Korzhnev DM.
Biomol NMR Assign. 2017;11(2):169–173.
doi: 10.1007/s12104-017-9741-z
PMID: 28265855
2016
Targeting translesion synthesis for cancer therapy
Korzhnev DM, Hadden MK.
J Med Chem. 2016;59(20):9321–9336.
doi: 10.1021/acs.jmedchem.6b00596
PMID: 27362876
2015
USP7–ICP0 interaction structure
Pozhidaeva AK, Mohni KN, Dhe-Paganon S, Arrowsmith CH, Weller SK, Korzhnev DM, Bezsonova I.
J Biol Chem. 2015;290(38):22907–22918.
doi: 10.1074/jbc.M115.664805
PMID: 26224631
HLTF HIRAN domain and fork reversal
Kile AC, Chavez DA, Bacal J, Eldirany S, Korzhnev DM, Bezsonova I, Eichman BF, Cimprich KA.
Mol Cell. 2015;58(6):1090–1100.
doi: 10.1016/j.molcel.2015.05.013
PMID: 26051180
2014
Rad18 zinc finger–ubiquitin structure
Rizzo AA, Salerno PE, Bezsonova I, Korzhnev DM.
Biochemistry. 2014;53(37):5895–5906.
doi: 10.1021/bi500823h
PMID: 25162118
2013
Rev1–PCNA interaction mapping
Pustovalova Y, Maciejewski MW, Korzhnev DM.
J Mol Biol. 2013;425(17):3091–3105.
doi: 10.1016/j.jmb.2013.05.029
PMID: 23747975
Loss of structure–gain of function (commentary)
Korzhnev DM.
J Mol Biol. 2013;425(1):17–18.
doi: 10.1016/j.jmb.2012.10.012
PMID: 23084974
2012
Rev1 C-terminal binding sites
Pustovalova Y, Bezsonova I, Korzhnev DM.
FEBS Lett. 2012;586(19):3051–3056.
doi: 10.1016/j.febslet.2012.07.021
PMID: 22828282
NMR structure of Rev1–Polη interaction
Pozhidaeva A, Pustovalova Y, D’Souza S, Bezsonova I, Walker GC, Korzhnev DM.
Biochemistry. 2012;51(27):5506–5520.
doi: 10.1021/bi300566z
PMID: 22691049
FF domain folding intermediate (PNAS)
Korzhnev DM, Religa TL, Kay LE.
PNAS. 2012;109(44):17777–17782.
doi: 10.1073/pnas.1201799109
PMID: 22647611
Cross-validation of folding intermediate structure
Barette J, Velyvis A, Religa TL, Korzhnev DM, Kay LE.
J Phys Chem B. 2012;116(23):6637–6644.
doi: 10.1021/jp209974f
PMID: 22148426
2011
FF domain folding intermediate structure
Korzhnev DM, Vernon RM, Religa TL, Hansen AL, Baker D, Fersht AR, Kay LE.
J Am Chem Soc. 2011;133(28):10974–10982.
doi: 10.1021/ja203686t
PMID: 21639149
2010
Copper-binding domains NMR study
Fatemi N, Korzhnev DM, Velyvis A, Sarkar B, Forman-Kay JD.
Biochemistry. 2010;49(39):8468–8477.
doi: 10.1021/bi1008535
PMID: 20799727
Chemical shift difference measurement method
Bouvignies G, Korzhnev DM, Neudecker P, Hansen DF, Cordes MH, Kay LE.
J Biomol NMR. 2010;47(2):135–141.
doi: 10.1007/s10858-010-9418-8
PMID: 20428928
R1ρ vs HSQC comparison
Auer R, Hansen DF, Neudecker P, Korzhnev DM, Muhandiram DR, Konrat R, Kay LE.
J Biomol NMR. 2010;46(3):205–216.
doi: 10.1007/s10858-009-9394-z
PMID: 20033258
2009
Ubiquitin–SH3 interaction dynamics
van Ingen H, Korzhnev DM, Kay LE.
J Phys Chem B. 2009;113(29):9968–9977.
doi: 10.1021/jp902793y
PMID: 19569643
2008
Protein folding and relaxation dispersion review
Korzhnev DM, Kay LE.
Acc Chem Res. 2008;41(3):442–451.
doi: 10.1021/ar700189y
PMID: 18275162
2007
MARK3 UBA instability
Murphy JM, Korzhnev DM, Ceccarelli DF, Briant DJ, Zarrine-Afsar A, Sicheri F, Kay LE, Pawson T.
PNAS. 2007;104(36):14336–14341.
doi: 10.1073/pnas.0703012104
PMID: 17726107
FF domain folding pathway
Korzhnev DM, Religa TL, Lundström P, Fersht AR, Kay LE.
J Mol Biol. 2007;372(2):497–512.
doi: 10.1016/j.jmb.2007.06.012
PMID: 17689561
Barnase dynamics propagation
Zhuravleva A, Korzhnev DM, Nolde SB, Kay LE, Arseniev AS, Billeter M, Orekhov VY.
J Mol Biol. 2007;367(4):1079–1092.
doi: 10.1016/j.jmb.2007.01.051
PMID: 17306298
2006
SH3 folding hydration and packing
Bezsonova I, Korzhnev DM, Prosser RS, Forman-Kay JD, Kay LE.
Biochemistry. 2006;45(15):4711–4719.
doi: 10.1021/bi060177r
PMID: 16605239
Fyn SH3 folding intermediates
Korzhnev DM, Neudecker P, Zarrine-Afsar A, Davidson AR, Kay LE.
Biochemistry. 2006;45(34):10175–10183.
doi: 10.1021/bi0611560
PMID: 16922492
SH3 relaxation dispersion analysis
Neudecker P, Korzhnev DM, Kay LE.
J Biomol NMR. 2006;34(3):129–135.
doi: 10.1007/s10858-006-0001-2
PMID: 16604422
2005
SH3 folding side-chain interactions
Mittermaier A, Korzhnev DM, Kay LE.
Biochemistry. 2005;44(47):15430–15436.
doi: 10.1021/bi051771o
PMID: 16300390
Enzyme intrinsic dynamics and catalysis
Eisenmesser EZ, Millet O, Labeikovsky W, Korzhnev DM, Wolf-Watz M, Bosco DA, Skalicky JJ, Kay LE, Kern D.
Nature. 2005;438(7064):117–121.
doi: 10.1038/nature04105
PMID: 16267559
Multiple-site exchange NMR dispersion
Korzhnev DM, Neudecker P, Mittermaier A, Orekhov VY, Kay LE.
J Am Chem Soc. 2005;127(44):15602–15611.
doi: 10.1021/ja054550e
PMID: 16262426
Cross-correlated spin relaxation effects
Korzhnev DM, Mittermaier AK, Kay LE.
J Biomol NMR. 2005;31(4):337–342.
doi: 10.1007/s10858-005-2468-7
PMID: 15929000
Off-resonance R1ρ dynamics
Korzhnev DM, Orekhov VY, Kay LE.
J Am Chem Soc. 2005;127(2):713–721.
doi: 10.1021/ja0446855
PMID: 15643897
2004
Azurin electron transfer dynamics
Zhuravleva AV, Korzhnev DM, Kupce E, Arseniev AS, Billeter M, Orekhov VY.
J Mol Biol. 2004;342(5):1599–1611.
doi: 10.1016/j.jmb.2004.08.001
PMID: 15364584
Fyn SH3 folding intermediates (Nature)
Korzhnev DM, Salvatella X, Vendruscolo M, Di Nardo AA, Davidson AR, Dobson CM, Kay LE.
Nature. 2004;430(6999):586–590.
doi: 10.1038/nature02655
PMID: 15282609
Multiple-quantum relaxation dispersion
Korzhnev DM, Kloiber K, Kay LE.
J Am Chem Soc. 2004;126(23):7320–7329.
doi: 10.1021/ja049968b
PMID: 15186169
Protein L7/L12 ribosome switching
Bocharov EV, Sobol AG, Pavlov KV, Korzhnev DM, Jaravine VA, Gudkov AT, Arseniev AS.
J Biol Chem. 2004;279(17):17697–17706.
doi: 10.1074/jbc.M313384200
PMID: 14960595
Double- and zero-quantum NMR dispersion
Orekhov VY, Korzhnev DM, Kay LE.
J Am Chem Soc. 2004;126(6):1886–1891.
doi: 10.1021/ja038620y
PMID: 14871121
Protein folding acceleration by nonnative stabilization
Di Nardo AA, Korzhnev DM, Stogios PJ, Zarrine-Afsar A, Kay LE, Davidson AR.
PNAS. 2004;101(21):7954–7959.
doi: 10.1073/pnas.0400550101
PMID: 15148398
Methyl-TROSY for large proteins
Korzhnev DM, Kloiber K, Kanelis V, Tugarinov V, Kay LE.
J Am Chem Soc. 2004;126(12):3964–3973.
doi: 10.1021/ja039587i
PMID: 15038751
2003
T4 lysozyme exchange dynamics
Korzhnev DM, Orekhov VY, Dahlquist FW, Kay LE.
J Biomol NMR. 2003;26(1):39–48.
doi: 10.1023/a:1023039902737
PMID: 12766401
Azurin low-populated states
Korzhnev DM, Karlsson BG, Orekhov VY, Billeter M.
Protein Sci. 2003;12(1):56–65.
doi: 10.1110/ps.0225403
PMID: 12493828
2002
TGF-β3 dynamics-modulated activity
Bocharov EV, Korzhnev DM, Blommers MJ, Arvinte T, Orekhov VY, Billeter M, Arseniev AS.
J Biol Chem. 2002;277(48):46273–46279.
doi: 10.1074/jbc.M206274200
PMID: 12221089
Heteronuclear spin-lock relaxation measurement
Korzhnev DM, Skrynnikov NR, Millet O, Torchia DA, Kay LE.
J Am Chem Soc. 2002;124(36):10743–10753.
doi: 10.1021/ja0204776
PMID: 12207529
2001–1995
Zervamicin backbone dynamics
Korzhnev DM, Bocharov EV, Zhuravlyova AV, Orekhov VY, Ovchinnikova TV, Billeter M, Arseniev AS.
FEBS Lett. 2001;495(1–2):52–55.
doi: 10.1016/s0014-5793(01)02363-8
PMID: 11322946
Off-resonance CPMG effects
Korzhnev DM, Tischenko EV, Arseniev AS.
J Biomol NMR. 2000;17(3):231–237.
doi: 10.1023/a:1008348827208
PMID: 10959630
Nanosecond dynamics and diffusion
Orekhov VY, Korzhnev DM, Pervushin KV, Hoffmann E, Arseniev AS.
J Biomol Struct Dyn. 1999;17(1):157–174.
doi: 10.1080/07391102.1999.10508348
PMID: 10496429
Model-free dynamics limitations
Korzhnev DM, Orekhov VY, Arseniev AS.
J Magn Reson. 1997;127(2):184–191.
doi: 10.1006/jmre.1997.1190
PMID: 9281482
Bacterioopsin backbone dynamics
Orekhov VY, Pervushin KV, Korzhnev DM, Arseniev AS.
J Biomol NMR. 1995;6(2):113–122.
doi: 10.1007/BF00211774
PMID: 22910799
Helical subunit dynamics in bacterioopsin
Pervushin KV, Orekhov VY, Korzhnev DM, Arseniev AS.
J Biomol NMR. 1995;5(4):383–396.
doi: 10.1007/BF00182282
PMID: 22911558
