The major focus of my research is cellular development of the cerebral cortex. The two main cell types found in the central nervous system, glia and neurons, are interconnected and we are equally interested in both cell types. We are particularly focused on molecular characteristics and cell fate determinants of progenitor cells, as this is important to create potential future therapies using these cells.
A large repertoire of cortical progenitor cells may in part explain the unique complexity of the human cerebral cortex. Radial glia cells (RGC) in the human cortical proliferative zones are multipotent progenitors that can generate all major neural cells (neurons, astrocytes and oligodendrocytes). We established organotypic slice cultures and dissociated cell cultures of the human fetal brains, which made experimental studies feasible. Using this approach, we would like to answer following questions: a) What determines the fate of cortical progenitor cells? b) What is the role of transcription factors such as Pax6, Nkx2.1and Olig1,2 in these processes? To study RGCs and their progeny, we transfect cells from cortical proliferative zone with plasmids or viruses that can specifically label RG cells. Progeny of labeled cell is identified with specific neuronal markers and studied electrophysiologically (in collaboration with Dr. Antic from our Department).
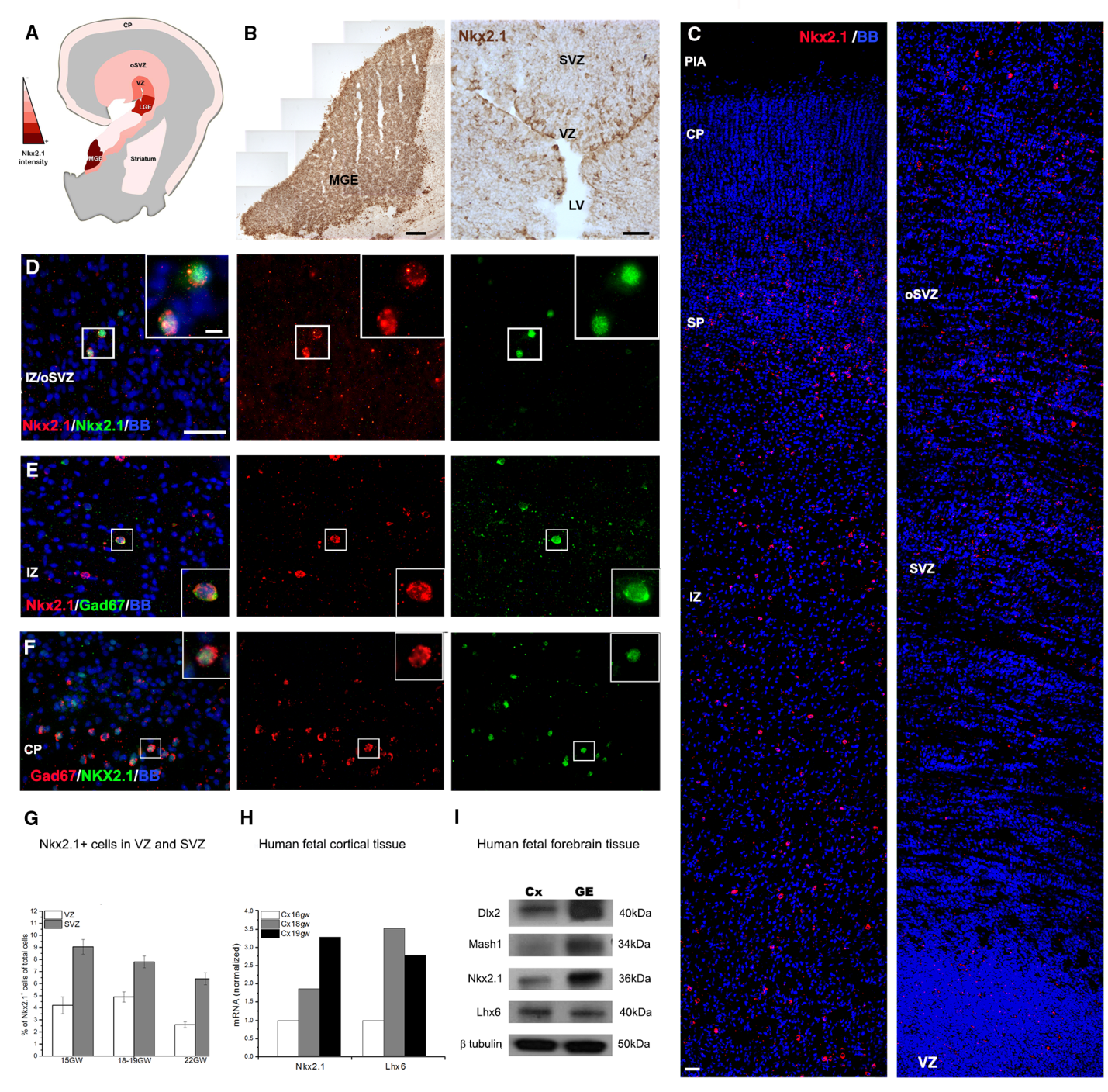
We study progenitors of cortical interneurons (GABAergic cells) by single and double-immunolabeling with cell-type specific markers combine with markers of cell proliferation (Ki67, BrdU). Recently we demonstrated the distribution of ventral transcription factor Nkx2.1, in the human fetal cerebral cortex at mid-gestation (20gw). This unexpected localization suggests local origin of a subtype of cortical interneurons. We determined that, unlike in rodents, cortical interneurons in the human fetal brain have dual origin. The significance of this finding is that these human-specific cortical interneurons that are generated in the cortex cannot be studied in animal models, and they might be involved in diseases such as schizophrenia or autism (Fig.3, Radonjic et al., Cell Reports 2014c; Selemon and N.Zecevic 2015).
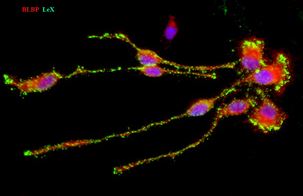
Another part of our research is to study oligodendrocyte lineage. Studies are being done mainly in human fetal forebrain sections (15-20 gestational weeks, gw) applying immunoflorescence. Parallel experiments are done in vitro and also in age-matched mice to evaluate species-specific differences.
In RGC cultures obtained from cortical proliferative zone at midgestation we study the effect of various environmental factors, such as sonic hedgehog (SHH) and Wnt signaling, hypoxia or NMDA receptor blokers on specification and differentiation of human cortical progenitors (Radonjic et al., 2014b, Ortega et al., 2013, 2016, Bagarsawala et al 2016a,b).